
Changes in BMD Testing in Ontario

Changes in BMD Testing in Ontario
Contributors: Dr. Adrian Lau, Dr. Heather McDonald-Blumer, Dr. Sandra Kim, Dr. Natasha Gakhal, Dr. Lianne Tile, Dr. Angela Cheung, Dr. Julie Gilmour, Dr. Laëtitia Michou, Dr. Claudia Gagnon, Dr. Zahra Bardai , Dr. Vithika Sivabalasundaram, Dr. Marie-Josée Bégin, Sarah Rydahl, Dr. Shirine Usmani, Dr. Alan Low, Dr. Joanna Sale, Dr. Teri Charrois, Dr. Laura Rothman, Dr. Jenny Thain, Irma Amri
The Ontario Health Insurance Plan (OHIP) Schedule of Benefits mandates health services reimbursement in the province of Ontario. As of April 1, 2026, significant changes were implemented, affecting access to Bone Mineral Density (BMD) testing in Ontario.
The information is posted on the Government of Ontario’s website under the OHIP Schedule of Benefits and fees.*
Osteoporosis Canada wants you to be aware of these changes as they will affect how you and your health care provider move forward when planning your bone health strategy.
Additionally, there may be a role for patient advocacy as the new intervals for bone density testing will restrict access to testing that may negatively affect patient care in some circumstances.
Osteoporosis Canada does not think that the current access as outlined by OHIP is consistent with the Osteoporosis Canada 2023 Clinical Practice Guideline for the Management of Osteoporosis in Canada and is not in the best interests of patient care.
Prior to April 1, 2026, physicians were provided with guidance from OHIP regarding the frequency of bone density testing in different clinical situations but were granted the opportunity to use their expertise to determine which patients were low risk (and therefore did not need frequent BMD monitoring) and which patients were of higher risk, benefiting from more frequent assessment. A repeat BMD was permitted 12 months after a previous test, if this was deemed necessary for high risk individuals by the health care provider.
As of April 1, 2026, OHIP stipulates that the ability to obtain a baseline BMD remains based on a healthcare provider’s assessment. However, the timing of the follow-up BMD is based on OHIP’s definition of patient risk. These new criteria used by OHIP are different from the generally accepted clinical risk factors used by health care providers across Canada, thereby limiting clinical appropriate access to necessary BMD testing for patients in Ontario.
For the purposes of subsequent testing, patient risk is defined using the fracture risk assessment tool (FRAX) or a recognized similar 10-year fracture prediction tool.
High risk is defined by OHIP as a patient with an estimated 10-year fracture risk of > 15%. This allows a patient to have their BMD monitored once every 36 months.
The only situation where a repeat BMD is permitted in 12 months, would be for individuals who have an endocrine condition known as Hypercortisolism/Cushing’s syndrome or individuals who are receiving high-dose glucocorticoid therapy, Prednisone (or equivalent) > 20 mg per day.
The new OHIP Schedule of Benefits references the 2023 Osteoporosis Canada Guideline in making these changes.
The 2023 Osteoporosis Canada Guideline does indeed recommend repeat BMD testing in 3 years for those who have initiated pharmacotherapy for osteoporosis, or those who have a 10-year fracture risk of > 15%.
However, a very important caveat that was not captured in the new OHIP Schedule of Benefits, is that BMD testing “should be reassessed earlier if patient develops secondary causes, new fracture or other risk factor for rapid bone loss”.

§Should be reassessed earlier if patient develops secondary causes (Appendix 1, Supplementary Table 5), new fracture or other risk factor for rapid bone loss.
These are the “secondary causes” found in the guideline’s supplementary table.
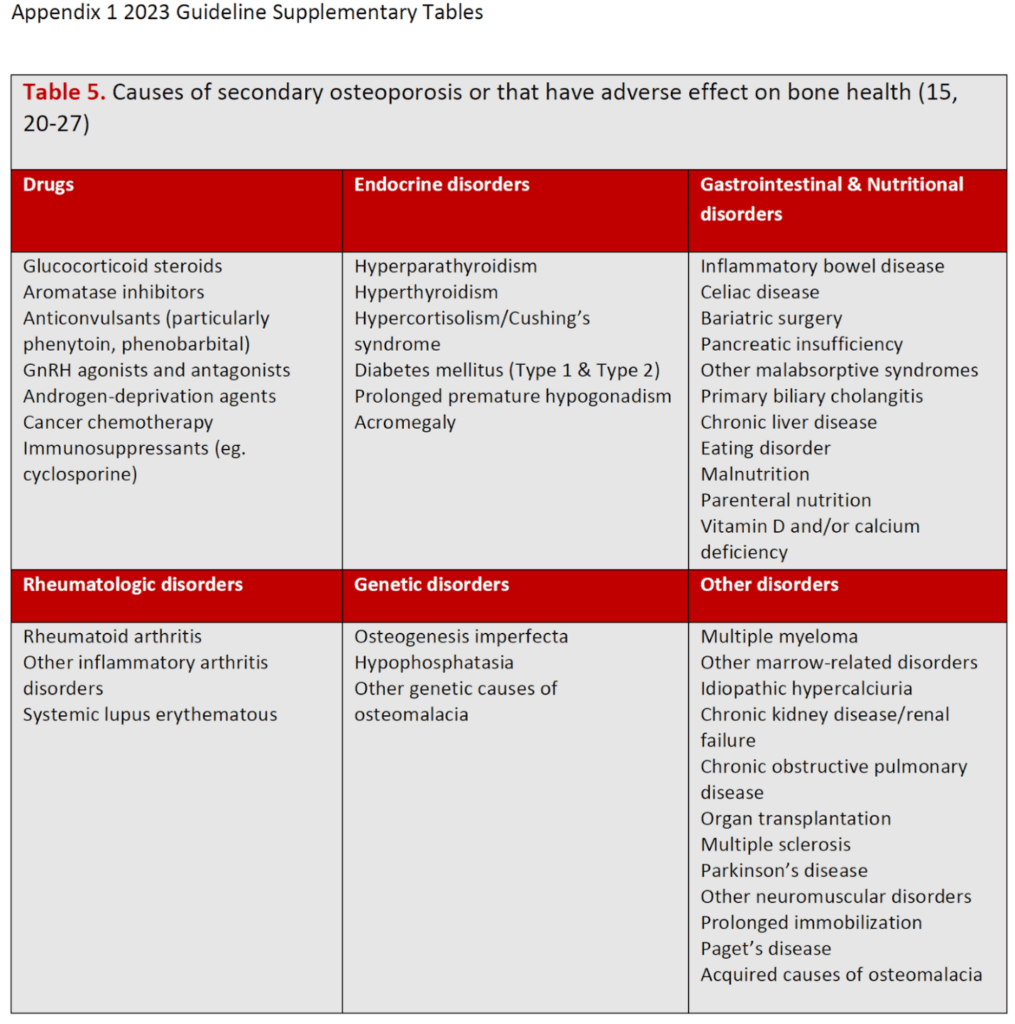
OHIP recognizes that those with “Hypercortisolism/Cushing’s syndrome or receiving high-dose glucocorticoid therapy of >20 mg Prednisone equivalent per day” may have a repeat BMD in 12 months but have neglected those who have other secondary causes for rapid bone loss. Of note, this dose of prednisone is higher than what the literature indicates is a threshold for bone loss and need of more frequent BMD monitoring.
For those who have other secondary causes (such as hyperparathyroidism, rheumatoid arthritis or genetic causes of metabolic bone disease), or those starting on medications associated with accelerated bone loss which are not glucocorticoids (such as aromatase inhibitors or androgen deprivation therapy) – these individuals will need to wait 36 months for a repeat BMD test. For those who have had a new fragility fracture, a repeat BMD suggested by the health care provider or Fracture Liaison Service (FLS) at the hospital may need to be deferred if a previous test was completed within the past 3 years. Those who are on pharmacotherapy for bone health, transitioning off anabolic / bone formation therapy or denosumab therapy may be at increased risk of rapid bone loss, and may benefit from repeat BMD testing earlier than 36 months.
Furthermore, after the introduction of these changes in April 2026, patients, healthcare providers, BMD testing centres, clinics, and hospitals were given little notice, and had little time to prepare for the implications.
Of note, the 2023 Osteoporosis Canada Guideline addresses postmenopausal females and males aged 50 years or older, and does not apply to those who are younger, including children with secondary osteoporosis and fractures. Due to their rate of bone growth, a repeat BMD earlier than 3 years may be beneficial.
Implications:
- For patients: Patients who genuinely need earlier monitoring are vulnerable to preventable and potentially significant bone loss that could have been identified and treated sooner. This may result in delays in addressing increasing fracture risk. They will go without essential testing or pay out of pocket.
- For doctors: Doctors lose the ability to monitor treatment response or disease progression appropriately, limiting their clinical judgment and forcing a one‑size‑fits‑all approach that may compromise care.
- BMD tests that are already booked and scheduled within the next 36 months may no longer be covered under the new OHIP guidelines. These tests may need to be cancelled or postponed.
- As the risk of osteoporosis and fragility fractures are higher in women, these changes have the greatest impact on women’s health.
Next steps:
- Osteoporosis Canada believes that repeat BMD testing in 12 months should be allowed beyond those with “Hypercortisolism/Cushing’s syndrome or receiving high-dose glucocorticoid therapy of >20 mg Prednisone equivalent per day”, and should include patients with other secondary causes of osteoporosis, new fractures, or other risk factors for rapid bone loss.
- Osteoporosis Canada understands that the sudden changes in access to BMD testing leaves patients, healthcare providers and clinics scrambling, as scheduled BMD tests may no longer be OHIP covered.
- Osteoporosis Canada understands that some patients will not qualify for repeat BMD testing until 36 months later, though an earlier reassessment may be necessary.
- Osteoporosis Canada is in support of deferring any changes to BMD testing policies until evidence-based expert advice is incorporated to support the OHIP policies, and to give all stakeholders more time to adapt their clinical practices to adhere to the new policies.
- Osteoporosis Canada is currently working with advocacy consultants towards a coordinated advocacy response strategy.
- Members of Osteoporosis Canada’s Scientific Advisory Council are in contact with the Ontario Medical Association and Ontario Association of Radiology to discuss advocacy efforts.
- A meeting with representatives from Ontario’s Ministry of Health is currently being arranged to discuss the above concerns.
References:
*OHIP Schedule of Benefits: https://www.ontario.ca/files/2026-03/moh-schedule-benefit-2026-03-27.pdf
Clinical practice guideline for management of osteoporosis and fracture prevention in Canada: 2023 update: https://www.cmaj.ca/content/195/39/E1333
Scientific Advisory Council
Osteoporosis Canada’s rapid response team, made up of members of the Scientific Advisory Council, creates position statements as news breaks regarding osteoporosis. The position statements are used to inform both the healthcare professional and the patient. The Scientific Advisory Council (SAC) is made up of experts in Osteoporosis and bone metabolism and is a volunteer membership.


